Taking steps to preserve fertility for men and women is important to the physicians and staff at GENESIS. To set our patients’ minds at ease, our laboratory director, Dr Alka Goyal, answers some pressing questions about the safety of your frozen eggs, sperm and embryos.

Why do we freeze embryos, eggs and sperm?
Embryo/Blastocyst freezing is a routine part of IVF and ICSI (intracytoplasmic sperm injection) treatment. It offers many benefits, not least the fact that embryos left over from a fresh cycle can be stored for use at a future date. Eggs / sperm / testicular tissue as well as embryos are also frozen to preserve fertility in case of disease / medical treatment and/or to delay child bearing.
How does GENESIS Fertility store frozen eggs, sperm, and embryos?
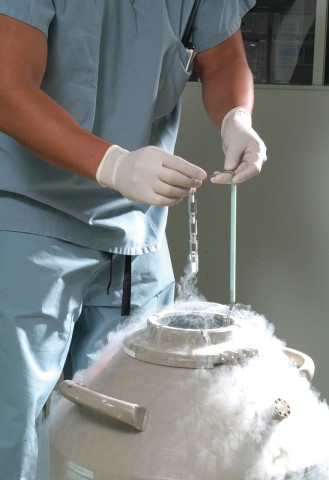
At GENESIS we use a freezing process known as Vitrification after which each specimen is loaded in straws (vials) with patients details and then stored in cryogenic tanks at a temperature of -196°C (-321°F). Virtually all biological activity is arrested at these temperatures, allowing samples to be preserved for months or years without much damage (Fahy et al). In order to preserve the integrity of the samples it is extremely important that the temperature is maintained throughout storage period as -130°C is the glass transition point of water below which ice crystals will form and damage the sample.
What safeguards does GENESIS Fertility use for storing frozen eggs, sperm and embryos?
The tanks that contain frozen specimens are monitored 7 days a week. Each tank gets a physical inspection daily, looking for problems or signs of problems. We monitor the liquid nitrogen level of each tank and replenish the liquid nitrogen as needed, as it evaporates at a slow rate. The storage area is locked at all times and access is limited to authorized personnel only.
We also have a sophisticated electronic tank monitoring which uses Wi-Fi enabled sensors to ensure that tanks maintain the required temperature. Probes attached to the tank that detect a rise in temperature as well as a drop in the level of liquid in the tank ensuring dual safety. Sensors are connected to an alarm system that will alert staff to an alarm condition outside of normal working hours as well.
The larger tanks are connected to an automatic liquid nitrogen manifold which is connected to two supply tanks which automatically start to fill the cryotanks when the levels start to fall. If the tanks are empty it also sounds an alarm.
The laboratory staff follows written standard operating procedures pertaining to the storage, maintenance, retrieval and transfer of frozen specimens to ensure cryogenic safety. The facility is also subject to routine inspections by CAP and the NYDOH.
How do I know my frozen eggs, sperm and embryos are safe?
24/7 Monitoring
GENESIS cryogenic storage tanks are continuously monitored 24/7 via local and remote access as mentioned above. If the temperature is not within the permissible limit, our lab director and staff are instantly alerted either in or out of the laboratory with alerts via emails as well as their cell phones by a specialized alert system. Additionally, the liquid level probes are placed in the upper level in our tanks, so in case the alarms go off overnight, the levels will not fall low enough to cause damage, as the specimens are suspended in the lowest level.
What if there’s a power fail or blackout?
The liquid nitrogen is independent of any source of power and can thus withstand power outages. GENESIS also has a gas powered generator (which is tested monthly) that immediately turns on and delivers power to ALL laboratory equipment; there is zero downtime between a power failure and generator response.
Our Lab Credentials 
GENESIS maintains and operates three state-of-the-art laboratories licensed by the New York State Department of Health (NYSDOH), the U.S. Federal Drug Administration (FDA) and is accredited by the College of American Pathologists (CAP). GENESIS is proud to be a member in good standing with the Society for Assisted Reproductive Technology (SART).
Our Lab Director
Alka Goyal, PhD, HCLD/CC is the Director of Laboratories at GENESIS. She holds a High Complexity Laboratory Director’s license and a Clinical Consultant certification from the American Board of Bioanalysis. Dr. Goyal has the NYSDOH lab director certification and has been involved in the field of reproductive medicine for over 25 years. She has also recently completed a Global Clinical Research Scholars program from Harvard Medical School, Boston that provides advanced training in healthcare research.
If you would like to learn more about GENESIS Fertility New York or are ready to schedule an appointment, please speak with one of our representatives at 929-605-5467.